Scientists identify molecules that can block novel coronavirus
HEALTH | JUNE 14, 2020:
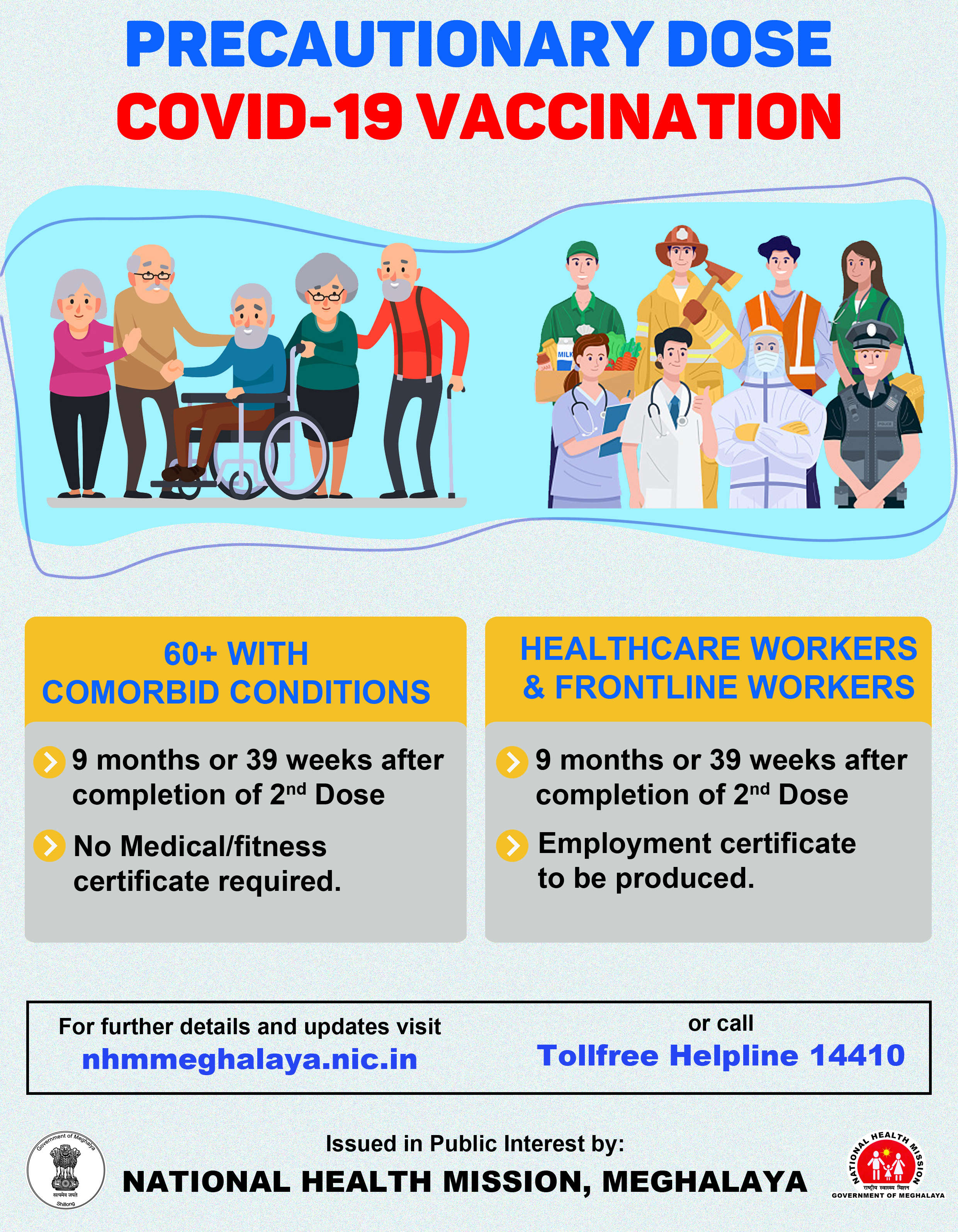
A team of scientists at the University of Georgia (UGA) in the US have identified a set of small molecules that can block the activity of a key protein in SARS-CoV-2 which is responsible for causing COVID-19.
The finding proves to be quite significant as it lights up a promising path for new therapeutics for the disease.
The team discovered that the SARS-CoV-2 protein PLpro is integral for the replication of the virus as well as its ability to suppress host immune function.
According to the research, which has been published in the journal ACS Infectious Diseases, the compounds, naphthalene-based PLpro inhibitors, are shown to be effective at halting SARS-CoV-2 PLpro activity as well as replication.
This led researchers to explore inhibitors designed to knock out PLpro to stop the virus from replicating and it all began with a series of compounds that were discovered 12 years ago.
The compounds were found to be effective against SARS but development was cut short since SARS had not reappeared.
Scott Pegan, a professor at UGA said, "Obviously now we see the current coronavirus is probably going to be with us for a while – if not this one, then probably other types of coronaviruses. Therefore, these compounds are a good starting point for therapeutic development."
"They have all the properties you would typically want to find in a drug, and they have a history of not being considered toxic," he added.
Meanwhile, researchers believe that the compounds offer a potential rapid development path to generating PLpro-targeted therapeutics for use against SARS-CoV-2.
"The kind of small molecules that we're developing are some of the first that is specifically designed for this coronavirus protease," Pegan said.
Pegan also informed that up till now, most therapeutic work against SARS has targeted another virulence factor, C3Lpro.
"This is a great start with a different target. We hope that we can turn this into a drug that we can get in front of the Food and Drug Administration," he said.
ALSO READ: